HYGIENIC VALVES & COMPONENTS
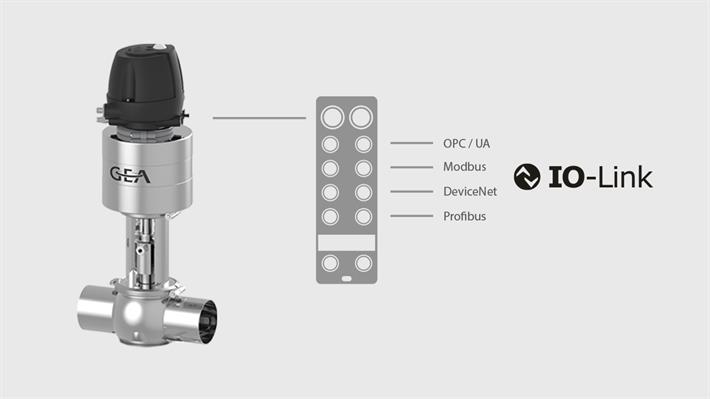
Hygienic valves from GEA are the key component as the core of fixed-pipe process plants. With a flexible valve concept and advanced control and automation functions, our valves offer the manufacturer maximum product safety and process reliability. All GEA hygienic valves are efficiently and economically designed for the respective application and enable sustainable operation with considerable savings potential. Complementary components in our program optimize the process plant – from pigging systems for recovering valuable products, process connections and compact expansion compensators for compensating thermal stresses to tank safety systems for securing and cleaning tanks and vessels. We regularly launch sophisticated product innovations on the market, supported by our research and development department. The market makes high demands, we meet them consistently and continuously.
Products
Let's stay in touch!
Contact us